Skeletal Muscle: Overview, Histology, and Pathology
Muscle tissue represents one of the four tissue in the human body with the epithelial tissue, connective tissue, and nervous tissue. Embryologically, the muscle tissue is derived by the mesoderm and it can be skeletal, cardiac, or smooth tissue type. The main differences between muscle tissue types rely on the histology, function, and location (Table 1) Skeletal Muscle Biopsy
Table 1. Comparison of Structure and Properties of Muscle Tissue Types.CC 3.0 BY OpenStax College, ‘Anatomy & Physiology, Connexions Web site http://cnx.org/content/col11496/1.6/, Jun 19, 2013
Skeletal muscles produce voluntary movement, locomotion, maintain posture, protect internal organs, generate heat, and are responsible for facial expression. The muscle cells are also called myocytes their numbers remain constant throughout life. The cells are multinucleated and organized in muscle fibers. The striated appearance is due to the regular alternation of actin and myosin (both contractile proteins of the sarcomere, the functional units of the skeletal muscle. Each skeletal muscle fiber is innervated by a motor neuron which activates the fiber to contract through the neuromuscular junction.
Muscle diseases, disorders, and myopathies [1, 2] can be due to
-Injuries (trauma, sports, or tumor excision)
-Lack of use (aging or disability)
-Fatigue
-Genetic condition: primary muscular dystrophies (Duchenne, Becker, facioscapulohumeral muscular dystrophy, myotonic dystrophy, myotonia congenita, mitochondrial myopathies)
-Autoimmune (myasthenia gravis, polymyositis, systemic sclerosis, rheumatoid arthritis)
-Toxins (toxic myopathies)
-Inflammation (bacterial or viral myositis)
-Cancer or myopathies associates such as dermatomyositis
-Endocrine myopathies (thyrotoxicosis, hyper- or hypo- thyroidism, Cushing syndrome, vitamin D deficiency)
-Periodic paralysis
Muscle biopsy – a tool for diagnosis and muscle disease evaluation
The clinical evaluation of muscle diseases includes clinical examination, electrodiagnostic, laboratory, and molecular genetic testing. The indication of muscle biopsy can be performed in the setting of quantifiable weakness and when the previous non-invasive clinical evaluation failed to identify a certain diagnosis in neuromuscular diseases [3]. The procedure for biopsy can be open or needle biopsy, depending on the incision (5-10 mm in the needle biopsy vs >30mm in the open biopsy) [4]. There are two major categories of histopathologic abnormalities observed in muscle disease, neurogenic atrophy, and myopathic changes. These structural changes often occur in tandem in myopathies [3]. The ideal amount of tissue depends on the use and the assessment technique: Skeletal Muscle Biopsy
-1 cm3 of frozen section for evaluating the morphology and enzyme histochemistry with light microscopy;
-5cm x 1cm of paraffin embedding tissue to evaluate the inflammation and the morphology of the inflamed cells;
-1-2 mm thick section for ultrastructural analysis with electron microscopy
-50-550 mg of tissue for biochemical testing such as for mitochondrial diseases.
Muscle biopsy beyond histology: preclinical models for pharmaceutical and toxicological studies and in regeneration medicine
Skeletal muscle tissue has a considerable capacity to regenerate thanks to the myoblasts’ activation, migration, proliferation, and fusion [5]. Myoblasts, the progenitor of skeletal muscle, are better surviving and proliferate in whole-muscle allograft than when isolated in cell culture, also in longer-term [6]. The interest of growing in vitro skeletal muscle has been studied as myoblast transfer therapy (MTT) for Duchenne muscular dystrophy exanimating the culture condition of major survival for myoblast based on effects of proteolytic enzymes, serum composition, and tissue preparation [7]. Ex vivo testing has been also suggested to study neuromuscular synapses by co-culturing spinal cord and muscle tissue, providing an adequate model system for toxic chemicals and antidotal treatment [8]. Skeletal Muscle Biopsy
A new protocol has been described on how to isolate human muscle progenitor cells (hMPCs) from skeletal muscle biopsy to address questions pertaining to skeletal muscle biology and regeneration [9]. This method has also a potential in regenerative medicine considering the limitation of the high donor heterogeneity.
Nowadays, muscle regeneration can be also be obtained from:
-Decellularized tissue by promoting cell homing, angiogenesis, and regeneration of myofibers and motor neuron axons [10].
-Skeletal muscle tissue engineering using polymers, such as poly(ethylene glycolic) (PEG), polyglycolic acid (PGA), poly(lactic-co-glycolic acid) (PLGA), poly(l-lactic acid) (PLLA), their copolymers (PLLA/PLGA), and polycaprolactone (PCL); natural polymers such as alginate, collagen, fibrin, or hyaluronic-acid, or a combination of the two [11-12] Skeletal Muscle Biopsy
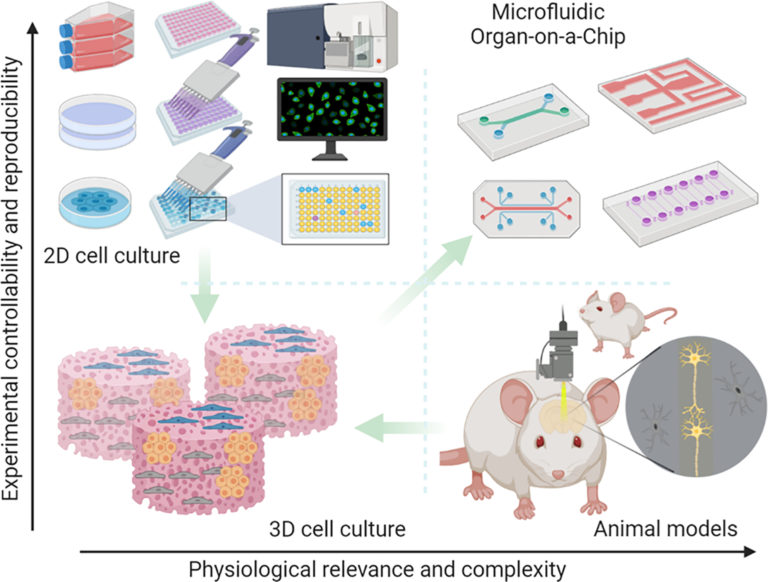
3D bioprinting has great potential in the engineering of functional 3D tissues in vitro for applications in drug discovery and regenerative medicine [13-14].
In a recent study of Gaurav Agrawal, engineered skeletal muscle tissue has been included in a skeletal muscle-on-a-chip[15]. The authors describe the architectural and structural complexities of muscle within a microfluidic device by characterizing the morphology and cells and tissue formation, useful as a potential alternative to animal testing in preclinical drug discovery. Further studies are requested to find adequate preclinical models for skeletal muscle diseases and treatments, also applying physiological gas and temperature conditions and fluidic flow.
References :
[1] Bradley WG, Henson RA et al, Muscle disease, Encyclopaedia Britannica, inc, September 2018, https://www.britannica.com/science/muscle-disease
[2] Agrawal, G., Aung, A., & Varghese, S. (2017). Skeletal muscle-on-a-chip: An in vitro model to evaluate tissue formation and injury. Lab on a Chip, 17(20), 3447–3461. https://doi.org/10.1039/c7lc00512a
[3] Joyce, N. C., Oskarsson, B., & Jin, L. (2015). Muscle Biopsy Evaluation in Neuromuscular Disorders Nanette. Phys Med Rehabil Clin N Am, 23(3), 609–631. https://doi.org/10.1016/j.pmr.2012.06.006.Muscle
[4] Meola G, Bugiardini E, Cardani R. Muscle biopsy. J Neurol. 2012 Apr; 259(4):601–10. [PubMed: 21805256]
[5] Lawson-Smith, M. J.; McGeachie, J. K. Experimental skeletal muscle grafts as a model of regeneration. Aust. N. Z. J. Surg. 67:35-39; 1997.
[6] Fan, Y.; Beilharz, M. W.; Grounds, M. D. A potential alternative strategy for myoblast transfer therapy: The use of sliced muscle grafts. Cell Transplant. 5:421-433; 1996
[7] Smythe, G. M., & Grounds, M. D. (2000). Exposure to tissue culture conditions can adversely affect myoblast behavior in vivo in whole muscle grafts: Implications for myoblast transfer therapy. Cell Transplantation, 9(3), 379–393. https://doi.org/10.1177/096368970000900309
[8] Drexler, B., Seeger, T., Grasshoff, C., Thiermann, H., & Antkowiak, B. (2011). Long-term evaluation of organophosphate toxicity and antidotal therapy in co-cultures of spinal cord and muscle tissue. Toxicology Letters, 206(1), 89–93. https://doi.org/10.1016/j.toxlet.2011.04.010
[9] Gheller, B. J., Blum, J., Soueid-Baumgarten, S., Bender, E., Cosgrove, B. D., & Thalacker-Mercer, A. (2019). Isolation, culture, characterization, and differentiation of human muscle progenitor cells from the skeletal muscle biopsy procedure. Journal of Visualized Experiments,2019(150), 1–10. https://doi.org/10.3791/59580
[10] Urciuolo, A., & De Coppi, P. (2018). Decellularized tissue for muscle regeneration. International Journal of Molecular Sciences, 19(8), 1–11. https://doi.org/10.3390/ijms19082392
[11] Saxena, A.K.; Marler, J.; Benvenuto, M.; Willital, G.H.; Vacanti, J.P. Skeletal muscle tissue engineering using isolated myoblasts on synthetic biodegradable polymers: Preliminary studies. Tissue Eng. 1999, 5, 525–532. [CrossRef] [PubMed]
[12] Page, R.L.; Malcuit, C.; Vilner, L.; Vojtic, I.; Shaw, S.; Hedblom, E.; Hu, J.; Pins, G.D.; Rolle, M.W.; Dominko, T. Restoration of Skeletal Muscle Defects with Adult Human Cells Delivered on Fibrin Microthreads. Tissue Eng. Part A 2011, 17, 2629–2640. [CrossRef] [PubMed]
[13] Laternser, S., Keller, H., Leupin, O., Rausch, M., Graf-Hausner, U., & Rimann, M. (2018). A Novel Microplate 3D Bioprinting Platform for the Engineering of Muscle and Tendon Tissues. SLAS Technology, 23(6), 599–613. https://doi.org/10.1177/2472630318776594
[14] Carosio, S., Barberi, L., Rizzuto, E., Nicoletti, C., Prete, Z. Del, & Musarò, A. (2013). Generation of eX vivo-vascularized Muscle Engineered Tissue (X-MET). Scientific Reports, 3, 1–9. https://doi.org/10.1038/srep01420
[15] Agrawal, G., Aung, A., & Varghese, S. (2017). Skeletal muscle-on-a-chip: An in vitro model to evaluate tissue formation and injury. Lab on a Chip, 17(20), 3447–3461. https://doi.org/10.1039/c7lc00512a


![Read more about the article Blood brain barrier microfluidic models [Brain on Chip]](https://innovation.cherrybiotech.com/staging/wp-content/uploads/2017/03/blood-brain-barrier-cubix-cherrybiotech-e1611761802407.jpg)