Introduction
The supporting vasculature is critical to the development of Adipose Tissue (AT) engineered models. Although it is essential for AT function and long-term maintenance, the Adipocyte-Endothelium Crosstalk is poorly understood.
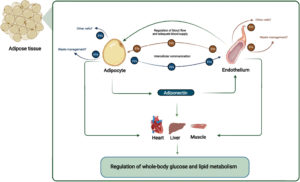
Because the crosstalk encompasses physiological adaptations such as blood flow alterations and molecular changes, the research relies largely on integrated models. In accordance with this, determining the precise function of Extracellular Vesicles in White Adipose Tissue biological control necessitates the development of novel experimental techniques as well as integrated models. Correlation studies are frequently used to infer the function of Extracellular Vesicles, while methods that inhibit exosome signaling in a cell-specific way are currently unavailable. Learn more through this review below by Sabaratnam et al.
Abstract
The authors state that “Obesity is characterized by pathological adipose tissue (AT) expansion. While healthy AT expansion enhances systemic insulin sensitivity, unhealthy AT expansion through increased adipocyte size is associated with insulin resistance, fibrosis, hypoxia, and reduced adipose-derived adiponectin secretion. The mechanisms causing the unhealthy AT expansion are not fully elucidated; yet, dysregulated crosstalk between cells within the AT is an important contributor.
Evidence from animal and human studies suggests a crucial role of the crosstalk between vascular endothelium (the innermost cell type in blood vessels) and adipocytes for metabolic homeostasis. Arterial endothelial cells are directly involved in maintaining normal organ functions through local blood flow regulation. The endothelial-dependent regulation of blood flow in AT is hampered in obesity, which negatively affects the adipocyte. Moreover, endothelial cells secrete Extracellular Vesicles (EVs) that target adipocytes in vivo.
The endothelial EVs secretion is hampered in obesity and may be affected by the adipocyte-derived adipokine adiponectin. Adiponectin targets the vascular endothelium, eliciting organ-protective functions through binding to T-cadherin. The reduced obesity-induced adiponectin binding of T-cadherin reduces endothelial EV secretion. This affects endothelial health and cell-cell communication between AT cells and distant organs, influencing systemic energy homeostasis.
This review focuses on the current understanding of endothelial and adipocyte crosstalk. We will discuss how obesity changes the AT environment and how these changes contribute to obesity-associated metabolic disease in humans. Particularly, we will describe and discuss the EV-dependent communication and regulation between adipocytes, adiponectin, and the endothelial cells regulating systemic energy homeostasis in health and metabolic disease in humans.”
Source
Sabaratnam R, Svenningsen P. Adipocyte-Endothelium Crosstalk in Obesity. Front Endocrinol (Lausanne). 2021 Aug 12;12:681290. doi: 10.3389/fendo.2021.681290. PMID: 34456860; PMCID: PMC8387580.