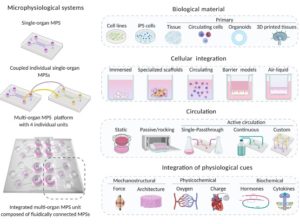
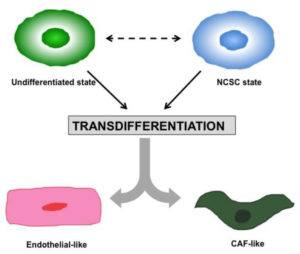
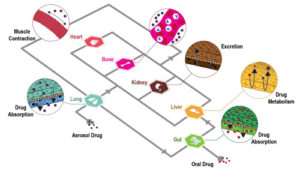
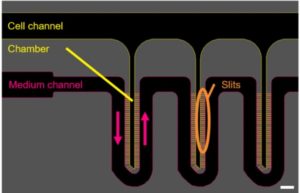
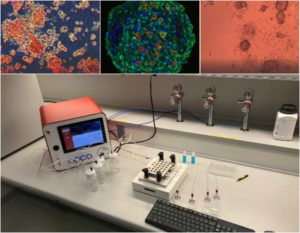
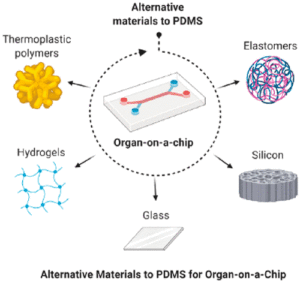
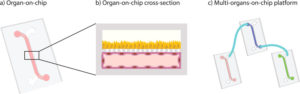
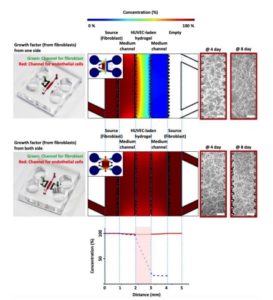
Introduction MPS (Multiorgan Microphysiological Systems) have evolved from tools to...
Learn more3D Vascularized Adipocytes Culture for obesity and Type 2 Diabetes Drug Development
Adipose Tissue Culture
Adipocytes, also known as lipocytes and fat cells, are the cells that primarily compose adipose tissue, specialized in storing energy as fat. Adipocytes are derived from mesenchymal stem cells which give rise to adipocytes through adipogenesis.
The adipocyte is important to the body in maintaining proper energy balance, storing calories in the form of lipids, mobilizing energy sources in response to hormonal stimulation, and commanding changes by signal secretions.
Under the microscope, the adipocyte appears bloated with triglycerides. The nucleus of the cell is displaced to one side by the fat. The cytoplasm of the cell looks like a thin line surrounding the pool of fat.
Can human adipose tissue be reproduced in a laboratory? It can now.
State of the art in 3D Adipocyte Culture and 3D Adipose Tissue
Researchers also established new 2D – accompanied by 3D – culture conditions from these human adipose stromal cells, making it possible to extract both adipocytes and endothelial cells from this tissue. The adipospheres obtained contained an intact vascular network arranged around adipocytes in the same way as the real human tissue. Better still, the adipocytes collected were able to distinguish between brown and white tissues (the two forms of human adipose tissue) (Brown Adipose Tissue and White Adipose Tissue) in the same manner as those present in the human body.
This breakthrough would also make it easier to research different medications that may be used to cure such disorders linked to adipose tissue anatomies, such as obesity or type 2 diabetes.
3D Vascularized Adipocytes Model
A reliable in vitro model of human adipose tissue may aid in the analysis of adipocyte function and provide improved resources for the screening of new therapeutic compounds for Type 2 Diabetes and other metabolic diseases. Cell culture models on two-dimensional surfaces stop short of imitating the 3D human in vivo adipose environments, whereas 3D culture models are often unable to sustain long-term cell culture due, in part, due to lack of physiomimitic microenvironmental control.
Microphysiological system (MPS) seems to be the most promising technology to accurately control the micro-environment of complex 3D biological models such as adipose tissue. A human psychomimetic models 3D vascularized model should integrate at least the 4 fundamental cell types of the adipose tissue: endothelial, macrophages, monocytes, and adipocytes.
This induces an additional level of complexity, in the control of the microenvironmental. Indeed, shear stress should be produce and maintain to sustain the endothelium differentiation, sufficient oxygen, and nutrient supply to maintain the culture over weeks, appropriate extracellular matrix stiffness, culture medium composition to maintain simultaneously 4 cellular types,…
Once achieved, MPS can mimic either a physiologic 3D vascularized adipose tissue (lipogenesis/lipolysis, adiponectin/leptin release, adipocytes maturation,…) or a physio-pathological condition. Indeed obesity mediated inflammation of the adipose tissue is one of the mechanisms leading to type 2 diabetes (free fatty acid release, macrophages activation, monocytes recruitment by endothelium,…).Hence, having an instrument able to maintain for the long term such a complex model in physiologic or pathological condition represents a much-needed tool in the drug development of type 2 diabetes, obesity, and other metabolic diseases therapies. Additionally, MPS represents a promising alternative to animal models, going in the direction of the 3R guidelines in Europe.
Melanoma Immunotherapy and Precision Medicine in the Era of Tumor Micro-Tissue Engineering: Where Are We Now and Where Are We Going?
Introduction Melanoma is clearly a complex disease with a high...
Learn moreMicrofluidic organ-on-chip system for multi-analyte monitoring of metabolites in 3D cell cultures
Introduction The researchers created a microfluidic organ-on-chip solution with integrated...
Learn moreA versatile microfluidic tool for the 3D culture of HepaRG cells seeded at various stages of differentiation
Introduction Animal models and two-dimensional (2D) cell cultures frequently fail...
Learn moreProbing Insulin Sensitivity with Metabolically Competent Human Stem Cell-Derived White Adipose Tissue Microphysiological Systems
Introduction Adipose Tissue is very versatile and essential for studies...
Learn morePumpless, modular, microphysiological systems enabling tunable perfusion for long-term cultivation of endothelialized lumens
Introduction The authors present a simple and modular Microphysiological system...
Learn moreFacilitating implementation of organs-on-chips by open platform technology
Introduction Many Organ-on-Chip platforms are still not robust for all...
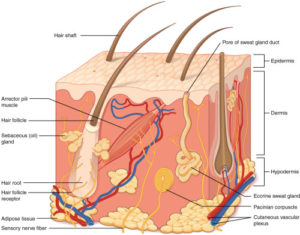
Learn moreSkin on a chip state of the art
Why a skin on a chip? The skin is the outer...
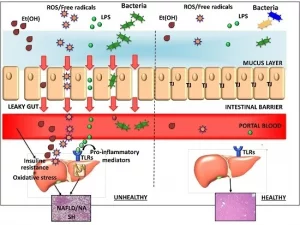
Learn moreThree-tissue microphysiological system for studying inflammatory responses in gut-liver Axis
Introduction The gut-liver axis is the bidirectional interaction that exists...
Learn moreLow-cost rapid prototyping and assembly of an open microfluidic device for a 3D vascularized organ-on-a-chip
Introduction In vitro generation of perfusable 3D microvessels is an...
Learn moreCo-culture With Human Breast Adipocytes Differentially Regulates Protein Abundance in Breast Cancer Cells
Introduction In postmenopausal women, fat mass and adipose-tissue mass (see...
Learn moreOrgan-on-Chips for Studying Tissue Barriers: Standard Techniques and a Novel Method for Including Porous Membranes Within Microfluidic Devices
Introduction Organ-on-Chips for Studying Tissue Barriers – Disruption of cell-formed...
Learn moreIsolation, Integration, and Culture of Human Mature Adipocytes Leveraging Organ-on-Chip Technology
Introduction Obesity and obesity-related co-morbidities are on the rise globally,...
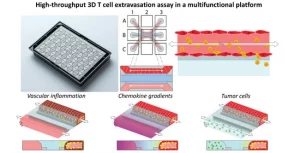
Learn moreA Microfluidic 3D Endothelium-on-a-Chip Model to Study Transendothelial Migration of T Cells in Health and Disease
Introduction Microfluidic 3D Endothelium-on-a-Chip – The endothelium is a thin...
Learn more3D Microphysiological System- Vascularized Tissue Constructs for Regenerative Medicine
3D Microphysiological System Vascularized Tissue – Human vascular microphysiological systems...
Learn more